There are no products listed under this category.
Estriol Benefits for Transition of Life
Estriol Benefits for the Transition of Life
Dr. Wulf Utian, the editor of the North American Menopause Society's journal, “Menopause”, stated, "Estriol has the potential of reduced risk, but similar benefits to alternative estrogen or estrogen-progestin formulations. The potential to minimize risks yet maximize most benefits places Estriol in a unique category among estrogens.”.
Restored Intimacy for the Transition in Life, Tearing, Itchiness, Dryness, Use Directly on Vaginal Tissue or Skin
European Doctors have used Estriol for 60 years SAFELY for dryness, tearing and itchiness. Estriol will not affect the size of the vagina, but will help with skin health.
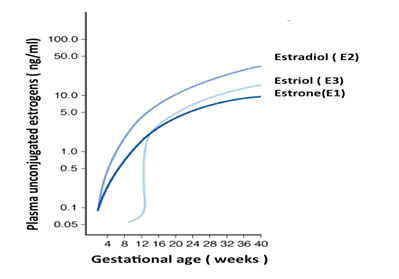
Estriol (E3) is the safest estrogen to take. Normally, the human female body produces very small amounts of Estriol. However, during the second and third trimesters of pregnancy Estriol production goes up 100 to 200 times. The woman produces 35mg-45mg of Estriol. The high levels of Estriol are credited with thickening and toughening up the skin of the birth canal. The birth canal skin is toughened up, so when the baby passes through the birth canal, the baby will not rip the skin of the birth canal. Women who are pregnant and deliver many babies statistically have a lower chance of breast cancer and endometrial cancer. Since high levels of Estriol are present during pregnancy, it is widely thought that Estriol is likely safe for baby and the mother.
Estriol Use in Actual Patient Testimonial for Feminine Health and Vitality
A Woman’s Body Produces 3 Types of Natural Estrogen: Estradiol, Estriol, and Estrone
A woman naturally produces 3 types of Estrogen. A woman produces estradiol, estriol, and estrone. A woman produces one kind of natural progesterone: Progesterone. Estradiol is a “Strong” estrogen. Estradiol is responsible for the formation of the characteristic woman’s breasts, hips, and butt. Too much Estradiol is associated with Breast Malignancy. In contrast, Estrone and Estriol are considered to be “Weak” estrogens. A “Weak” estrogen will go into the estrogen receptor and block a “Strong” estrogen from stimulating the estrogen receptor. Doctors have been backing away from Estrone because Estrone may be associated with Breast Malignancy. Also, looking at the corticosteroid synthesis chart, Estrone and Estradiol can change between each other. Both Estradiol and Estrone is associated with Breast Malignancy.
In contrast, Estriol canNOT change to Estradiol or Estrone or Cortisol. Estriol is a “Weak” Estrogen. Estriol is credited with blocking the Estrogen Receptor from “Strong” Estradiol. Estriol goes high during the 2nd and 3rd trimesters of pregnancy. Estriol stops the boy babies from looking like girl babies. Estriol blocks the Estrogen receptor in the boy from the mother’s Estradiol. Without Estriol the boy baby would end up looking like a girl. Without Estriol, the girl baby would develop breasts and a period. Estriol ensures that both babies look kind of alike. Both girl and boy babies look like babies and not women because Estriol blocks the mother’s Estradiol from affecting the babies.
Estriol is the Safest Estrogen to Take from Large Peer Reviewed Random Controlled Trials
Highly Significant, Large, Peer Reviewed Gold Standard Trials demonstrate that transdermal skin applied vaginal Estriol does not increase the risk of Breast Cancer:
1. Million Women’s Trial RR 0.67 [1]
2. Fournier RR 0.7[2]
3. Fournier[3,4]
4. Rosenberg [5]
5. Weiderpass [6]
Researcher Rosenburg showed that local transdermal Estriol had systemic effects. Local applied Estriol was NOT associated with any type of Breast Cancer in his Study.[5] Weiderpass found that oral Estriol and NOT topical vaginal Estriol may be associated with endometrial cancer. Weiderpass thought that progesterone may be given along with oral Estriol to decrease the risk of Endometrial Cancer.[6]
Eminent Hormone Supplement Researcher, Dr. Robert Greenblatt, in 1980, simply stated that Estriol relieved Hot Flashes and prevented Vaginal Dryness with no side effects. The editor of the North American Menopause Society’s Journal, “Menopause”, declared, "Estriol has the potential of reduced risk, but similar benefits to alternative estrogen or estrogen-progestin formulations. The potential to minimize risks yet maximize most benefits places Estriol in a unique category among estrogens.” Dr. Lauritzen showed that Estriol stopped Menopausal Discomfort in a 5 year clinical trial of oral Estriol Succinate in 940 Post Menopausal Women. Estriol protected against Breast Cancer, and Endometrial Cancer. In the 5 year clinical trial, 2 Breast Cancers developed (3 expected). In the 5 year trial, no Endometrial Cancer developed (3 expected).
Dr. Lemon in a clinical trial of 28 beast cancer patients used oral Estriol of 2.5-5 mg/day. Estriol induced remission in 6 of the women (37%). In the 1975 article in “Cancer Research”, Lemon, MD suggested that 2.5-5 mg/day of Estriol may be a recommended minimum dose of Estriol to prevent breast cancer. Pregnant woman typically excrete 30 - 50 mg/day of Estriol. The topical dose of Estriol is 0.5-1.0 mg/every other day because anything on the skin is 10 times the dose in potency compared to a topical transdermal delivery dose. Anything taken orally is 90% 1st pass inactivated by the liver.
Mainstream MD’s use Estriol to Help with Vaginal Atrophy - Itchy, Dryness, Skin Tearing - Restore Intimacy
Estriol will not help for vaginal shrinkage. Estriol helps for thickening up the vaginal skin to prevent tearing. Estriol helps for dryness and itchiness.
Mainstream MD’s treat vaginal atrophy using Estriol Cream. Many times after Menopause, half of the women have their vagina shrink both in circumference and length. Sometimes this makes penetration difficult. The vaginal skin bleeds many times after intercourse making intercourse painful and difficult. Estriol levels go up 100 times during the second and third trimester of pregnancy. Estriol are produced as high as 35-45 mg/day by the placenta. Libido goes high. Estriol thickens up the skin of the birth canal so that the baby can pass through the birth canal without ripping the skin of the birth canal. The recommended dose for topical Estriol for a post menopausal women is 0.5mg-1mg/every other day. It is suggested to apply the topical Estriol every other day because the wash out time of the Estriol is slightly longer than progesterone. The use of Estriol every day may lead to an unhealthy build up of Estriol. If this is the first time the patient has used Estriol, the patient may want to use Estriol for 10 continuous days to build up a level of Estriol. Topical transdermal progesterone may be used every day. Patients can use both 3 out 4 weeks. Patients take a 1 week Holiday to allow the progesterone and estriol receptors a rest. If the patient took both hormones continuously, then this may allow the hormone receptor sensitivity to decrease because of Down Regulation. Taking hormones continuously may make you less sensitive to hormones. The week long Holiday will prevent the patient from becoming less sensitive to both the Estriol and Progesterone.
Some patients that have strenuously eliminated all endocrine disruptors in their environment, can take Estriol and Progesterone continuously and slowly ramp down the supplement hormones over a period of 6 months to a year. These patients can completely eliminate hormone supplementation and still have healthy vaginal tissue and no Hot Flashes and no Night Sweats.
Horrific Night Sweats and Hot Flashes of Stressed Out Patients
Some patients that have high fear/stress/anxiety levels have horrific Night Sweats and Hot Flashes. Blood level Estradiol levels are very low. Sometimes I have even seen levels of less than 5 pg/gm. The normal blood estradiol level would be 60-100 pg/gm. Fear/stress/anxiety causes the patient to produce cortisol to deal with the stress. Because the patient is producing so much cortisol, the raw materials usually used to produce estradiol go into making cortisol. Thus, the patient’s estradiol is low. Because the estradiol is so low, the patient has tremendous Night Sweats and Hot Flashes. If progesterone is given to this stressed patient, the patient makes cortisol out of it, and the Hot Flashes are still horrible.
The solution is to give the patient Estriol. Estriol is not converted to cortisol, and so works to decrease Hot Flashes and Night Sweats directly. The normal topical dose for Estriol is 0.5-1.0 mg/every other day. However, for these stressed out patients, a dose of 2 mg/every other day may be more appropriate. Some practitioners also use ashweganda to decrease the cortisol being produced.
Urinary Tract Infections after Menopause Fought by Estriol
The vaginal lining after Menopause in the presence of “Franken Estrogens” (xenoestrogens) tends to degenerate. This degeneration of the vagina causes urinary tract infections (UTIs). In a 1993 New England Journal of Medicine study from Israel, 50 women were treated with 0.5 mg of Estriol Cream for 2 weeks. Then, these women took Estriol Cream twice a week for 8 months. 43 women had a placebo. The 50 women had 10 times less UTIs than the women that took placebos. The 50 women only had rare side effects.
A 1994 Swedish Study, 0.5 mg Estriol taken vaginally had equal effectiveness as a low dose estradiol vaginal ring for vaginal atrophy in menopausal women. Estriol led to improvements of vaginal dryness, painful intercourse, pain on urination, and urinary urgency. Estriol delivers these benefits without the side effect of Candida infections. In contrast, 12 women taking Estradiol had Candida Yeast infections.
Bone Loss Reversed by Estriol
In 1996, 3 Japanese Studies, an Italian Study, and a Taiwanese Study used Estriol to stop Bone Loss. All 3 Japanese Studies showed that 2mg/day of oral Estriol increased bone density 1-5% over the course of a year. The Italian Study demonstrated that 0.5 mg/day of vaginal Estriol Cream was able to somewhat prevent bone loss. The Taiwanese Study showed that Oral Estriol Succinate did not improve bone density. Topical transdermal Estriol Cream was shown to be more effective at increasing blood levels than Estriol taken orally. Most women were relieved of Night Sweats and Hot Flashes. No endometrial hyperplasia, the harbinger of endometrial cancer, was observed. Breakthrough bleeding was less.
No Increase in Blood Clots with Estriol
1 out of 5,000 women taking Estradiol and Estrone get blood clots. Wonderfully, Estriol does NOT increase blood clots. Estriol taken 8mg/day did not increase clotting factors in women. This is the consensus view of 1980’s of the review committee to review the efficacy of Estriol.
Less Wrinkles and Softer Skin
A 1996 Study from the “International Journal of Dermatology” measured skin vascularization, firmness, elasticity, moisture content, wrinkle depth, and pore size. 73%-100% of women using Estriol Cream over a period of 6 months showed improvement. Skin biopsy revealed type III collagen increase. The “Pregnancy Glow” of a woman is likely due to Estriol.
Treat Acne Scars
A 1995 article in “International Journal of Dermatology” demonstrated effective acne scar treatment with Estriol Cream.
Increase Sex Drive, Libido by Avoiding Xenoestrogens and Estriol and Progesterone
Sex Drive or Libido is killed by endocrine disruptors, known by the common name as xenoestrogens, “Franken Estrogens”. If you remove xenoestrogens completely, then usually, the sex drive comes back. Both Progesterone and Estriol go to extremely high levels during the second and third trimesters. A woman’s Sex Drive goes sky high during the second and third trimester. Progesterone causes an increase in Sex Drive. Estriol causes an increase in Sex Drive. Furthermore, Estriol will block all the “Franken Estrogens” that destroys the normal Sex Drive of the Woman. Progesterone levels go up 20 times. Estriol levels go up 250 times. [7, 8, 9]
To Restore a woman’s Sex Drive, it is very simple:
1. Remove all Xenoestrogens strenuously, most of the time this is all that is needed.
2. Take Progesterone
3. Take Estriol
Estriol Blood Levels in Pregnancy
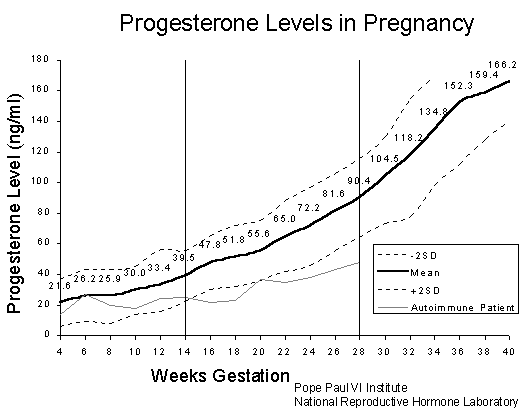
Progesterone Blood Levels in Pregnancy
Review Article for Estriol for the Academic
Here is a scholarly article that summarizes information about Estriol and its Clinical Use.
Notes
1. www.millionwomenstudy.org
2. Fournier A, Berrino F, Riboli E, Avenel V & Clavel-Chapelon F 2005 Breast cancer risk in relation to different types of hormone replacement therapy in the E3N-EPIC cohort. International Journal of Cancer 114 448–454. (https://doi.org/10.1002/ijc.20710)
3. Fournier A, Berrino F & Clavel-Chapelon F 2008a Unequal risks for breast cancer associated with different hormone replacement therapies: results from the E3N cohort study. Breast Cancer Research and Treatment 107 103–111. (https://doi.org/10.1007/ s10549-007-9523-x)
4. Fournier A, Fabre A, Mesrine S, Boutron-Ruault M-C, Berrino F & ClavelChapelon F 2008b Use of different postmenopausal hormone therapies and risk of histology- and hormone receptor-defined invasive breast cancer. Journal of Clinical Oncology 26 1260–1268. (https://doi.org/10.1200/JCO.2007.13.4338)
5. Rosenberg, L.U., Magnusson, C., Lindström, E. et al. Menopausal hormone therapy and other breast cancer risk factors in relation to the risk of different histological subtypes of breast cancer: a case-control study. Breast Cancer Res 8, R11 (2006). https://doi.org/10.1186/bcr1378
6. Weiderpass E, Baron JA, Adami HO, Magnusson C, Lindgren A, Bergstrom R, Correia N, Persson I: Low-potency oestrogen and risk of endometrial cancer: a case-control study. Lancet. 1999, 353: 1824-1828. 10.1016/S0140-6736(98)10233-7.
7. https://www.johnleemd.com/bioidentical-hormones.html - explains that 66% of women up to the age of 80 don’t need hormone replacement.
8. John Lee, MD, David Zava, Ph.D., and Virginia Hopkins, "What Your Doctor May NOT Tell You About Breast Cancer How Hormone Balance Can Help Save Your Life,” Hachette Book Group, New York, NY, 2002 pp. 153-171.
9. https://www.oatext.com/Estriol-review-Clinical-applications-and-potential-biomedical-importance.php Falah N, Torday J, Quinney SK, Haas DM (2015). Estriol review: Clinical applications and potential biomedical importance. Clin Res Trials 1: doi: 10.15761/CRT.1000109